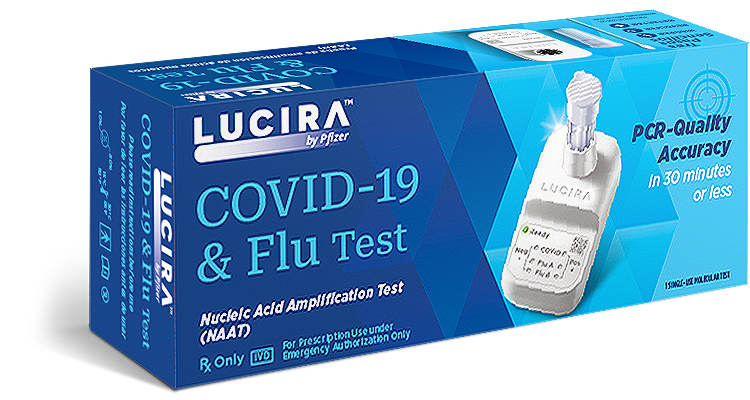
Lucira Covid-19 & Flu Test
Lucira Covid-19 & Flu Test NDC 00069970131
Single-use, molecular COVID-19 & flu A/B testing for your practice.
(for ages 2 and up)
Indication For Use
The LUCIRA® by Pfizer COVID-19 & Flu Test is authorized for the simultaneous qualitative detection and differentiation of SARS-CoV-2, Influenza A, and Influenza B viral RNA in anterior nasal swab specimens collected from individuals (2 years of age or older) who are suspected of respiratory viral infection consistent with COVID-19 by their healthcare provider. Emergency use of this test is limited to authorized laboratories. Testing is limited to laboratories certified under the Clinical Laboratory Improvement Amendments of 1988 (CLIA), 42 U.S.C. §263a, that meet requirements to perform high, moderate, or waived complexity tests. This test is authorized for use at the Point of Care (POC), i.e., in patient care settings operating under a CLIA Certificate of Waiver, Certificate of Compliance, or Certificate of Accreditation.
Non-Returnable