Orasure Quickflu Rapid Flu A & B Test, 22 Tests Box # 1001-0320
Orasure Quickflu Rapid Flu A & B Test, 22 Tests/Bx
# 1001-0320
Non-Returnable
The OraQuick® HCV test is FDA approved for detecting HCV antibodies in fingerstick and venipuncture whole blood. Our simple platform enables healthcare providers to deliver an accurate diagnosis in 20 minutes.
SENSITIVITY TO MEET YOUR NEEDS
OraSure QuickFlu® is an FDA cleared, CLIA-waived in vitro rapid qualitative test for the detection and differentiation of influenza type A and type B. An easy nasal swab procedure provides accurate results in as little as 10 minutes.
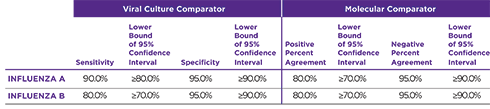
Orasure QuickFlu® delivers highly accurate results for flu types A and B and meets FDA’s new standards of performance.
- Proven analytical sensitivity across 51 different flu strains including CDC’s 2019-2020 flu strain panels
- Exceptional correlation to culture/PCR
PROVEN PERFORMANCE THAT MEETS FDA’S RECLASSIFICATION RULES
OraSure QuickFlu® exceeds the FDA’s reclassification requirement minimums for RIDTs.
- As of February 13, 2017, all RIDTs must meet the minimum acceptance performance criteria and comparator method for antigen based RIDTs. Tests that do not meet the new requirements cannot be used.
- Analytical reactivity testing must be performed on contemporary influenza strains and newly emerging strains in the event of an emergency.
ENSURES RESPONSIVE STANDARD OF CARE
- Simple, easy-to-use procedure with built-in control line
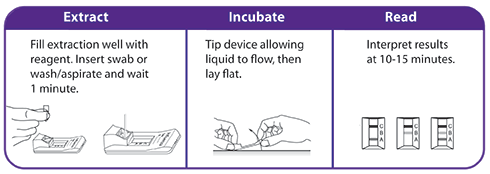
- Includes a flocked swab that optimizes sample collection and release resulting in superior performance
- American Society of Microbiology says, “NP flocked swabs collect better samples than routine NP swabs”
- Nasal swab collection – comfortable for the patient and simple for the HCP with no ancillary supplies required
- Ready-to-use reagents with room temperature storage
- Rapid testing that maximizes results
TEST AT THE POINT OF CARE
OraSure QuickFlu® is ideal for testing at the point of care in…
- Laboratories
- Emergency Rooms
- Physician Offices
- Public Health Facilities and Clinics
REIMBURSEMENT INFORMATION
CPT Codes
87804 QW- Influenza A Detection
87804-59- Influenza B Detection