Pfizer / Hospira
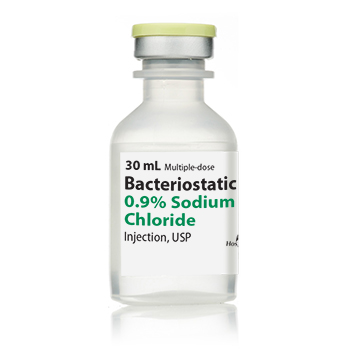
BACTERIOSTATIC 0.9% SODIUM CHLORIDE INJ, USP, MDV, 30ML, 25/PACK NDC 00409196607 NON-RETURNABLE
SKU030339577000
NDC00409-1966-07
Manufacturer Number00409196607
Application Diluent
Strength: 0.9%
Multiple Dose Vial
Storage Requirements USP Controlled Room Temperature
Type Intramuscular, Intravenous, or Subcutaneous
Volume 30 mL / PACK OF 25 VIALS
Latex Information indicates that natural rubber latex has not been used in the manufacture of this device or drug container closure system.